Clay minerals are ubiquitous on Earth surface. They are major components in many soil types, and are the main components of extended sedimentary stratigraphic layers.
Illite and smectite alone may constitute 30 w% of all sedimentary rocks.
The nano- to micrometer size of clay minerals particles, combined with their layered structures, provide them with high specific surface area
values, among the highest known for natural materials, and are responsible for their high adsorption capacities
for a range of chemicals spanning from natural organic matter to emerging organic and inorganic
pollutants.
The accuracy of predictions of geochemical interactions in many surficial and subsurface environments is thus partly dependent on our capability to consider
the various aspects of clay mineral reactivity in our modeling approaches. Clay mineral layers are negatively charged as a result of heterovalent cations structural substitutions.
This property is responsible for their well-known cation exchange capacity, but it is also responsible for specific electrostatic properties influencing the transfer of chemical species in the nearby porosity in a non-classical (meaning non-Darcian and non-Fickian) way.
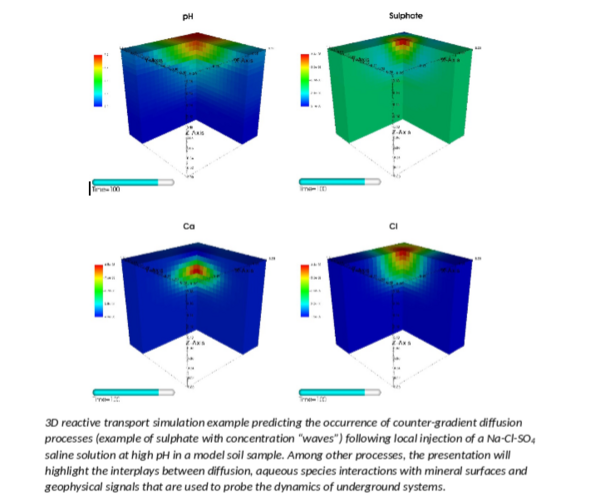
The prediction of mass transfers in natural clayey environments is thus made very difficult because of strong couplings between various chemical reactive properties and complex transport properties. In this presentation, we will show how newly developed reactive transport modeling capabilities in the code CrunchClay could help overcoming these difficulties with the prediction of geochemical as well as geophysical signals that can be monitored in clayey environments.